

|
|
|
#1
|
|||
|
|||
|
Why does my chiller affect my pH?
Every time the chiller comes on I experience a drop in my pH - why is that?

|
|
#2
|
|||
|
|||
|
My first thought would be electrical interference, but the correlation might be indirect. Interesting question.
__________________
Jonathan Bertoni |
|
#3
|
|||
|
|||
|
Yeah, there's someting funny going on. I'm not convinced the drop in pH is necessarily real. If we were just causing cooling (say toss in a bottle of ice) we'd see a very slight rise in pH.
__________________
FSM ~ Touched by His noodly appendage ~ |
|
#4
|
|||
|
|||
|
The change in temperature (lowering it to a cooler temperature) is affecting the pH of your tank. In general, all gases (oxygen, nitrogen, carbon dioxide) are more soluble at lower temperature in water.
For oxygen and nitrogen this is important as oxygen is more soluble at cooler temperatures, so more is available to fish. Think about how much more flow and aeration a tank needs when it begins to get hot and the fish being to respire rapidly. Carbon dioxide presents a twist on gas solublity in water. The amount of gas in solution is directly proportional to its concentration in the surrounding environment. This is known as Henry's Law in chemistry. This assumes that the gas does not react with the solvent (water in our case). This is true for oxygen and nitrogen, but not for carbon dioxide (shown below): CO2 + H2O <---> H2CO3 carbon dioxide + water equals carbonic acid, with the <---> arrow meaning they are in equilibrium with one another. So as you decrease the temperature of your water, even though it is over such a small range (only 1 degree Celcius), this affect the solubility of carbon dioxide, making is more soluble and it reacts with the abundant water to produce some carbonic acid. This then reacts to produce acid (H+) which lowers your pH and forms bicarbonate (HCO3-, a buffer we are familiar with). H2CO3 <----> H+ + HCO3- From the graph you've provided the later day "spikes" in temperature show an increase in pH, followed by a lowering when the chiller kicks in and again reduces the temperature. HTH, ChemLife |
|
#5
|
|||
|
|||
|
So you are suggesting that a 1 C difference in temperature is inducing enough CO2 to be taken up to reduce pH by 0.1 units? I think not

__________________
FSM ~ Touched by His noodly appendage ~ |
|
#6
|
|||
|
|||
|
Impressive first post ChemLife

|
|
#7
|
|||
|
|||
|
Guys I dunno where you get 1C - it is 0.5C....
|
|
#8
|
|||
|
|||
|
From 27.25 to 26.25 C--that's 1 C

__________________
FSM ~ Touched by His noodly appendage ~ |
|
#9
|
|||
|
|||
|
Nope - you are not reading correctly
 I'll annotate.... I'll annotate....The Temperature drop you need to look at is that which is caused by the chiller - as only during that short time there is a correlation between chiller = on and pH drop. The rest of the time has no bearing on the issue at hand. 
|
|
#10
|
|||
|
|||
|
Ha, ok. The initial big change in temp is I guess what jumps out. Either way same answer: there's got to be something "funny" going on here. I wouldn't be surprised if you're getting electrical interference with the meter or something like that.
Is water constantly flowing through the chiller, even when not actively chilling? cj
__________________
FSM ~ Touched by His noodly appendage ~ |
|
#11
|
|||
|
|||
|
Yup. the chiller is on my Aquatronica - controlled by an external temperature sensor. So the Aquatronica turns it on and off. The return pump feeds through the chiller to the main display. The pH probe is almost in front of the return pipe.
|
|
#12
|
|||
|
|||
|
That was a well done post by ChemLife and I agree.
A temp drop of 1 C will cause a shift in the pK1 and pK2 enough to cause an off-set in the CO2 to raise it from for 0.60 mg / l to .78 mg l if dealing with Normal std seawater. That off-set is enough to drop the pH 0.1 pH units. And if there was no temp shift, just to make a point here, where there is no shift in the pK1 and pK2 and the pH dropped from a 0.18 mg / lCO2 increase, you get the same drop in pH of~ 0.1 pH units. This is all based on Lyman pK1 , pK2 an pHnbs. Even if you went the more correct route, as pK1, pK2, pHsws the offest is about the same.
__________________
If you See Me Running You Better Catch-Up An explosion can be defined as a loud noise, accompanied by the sudden going away of things, from a place where they use to be. |
|
#13
|
|||
|
|||
|
Ok I am impressed by the pK1 and pHnbs but I still don't understand. The temperature drop was a little less than 0.5C. The corresponding pH drop was roughly 0.12 units.
I guess if I knew what pK1 and pK2 was .... |
|
#14
|
|||
|
|||
|
Lets put it this way - Boomer, do you agree with what ChemLife said? If so - then I understand. I know what
CO2 + H2O <---> H2CO3 means, but not what pK1 , pK2 an pHnbs means. Sometimes you are just too advanced for me 
|
|
#15
|
|||
|
|||
|
Thanks, ChemLife and Boomer!
__________________
Jonathan Bertoni |
|
#16
|
|||
|
|||
|
Yes I agree with ChemLife
 pK1 , pK2 an pHnbs and temp, salinity and Alk These can fitted into a series of equations worked by Buch and Park many year ago. So, in short form, if you know two of the 3 variables, pH, Alk and CO2 you can calculate the unknown . For fresh water only you can kinda' see how this works in this calculator, although temp is not inputed into this calculator . http://www.csd.net/~cgadd/aqua/art_plant_co2chart.htm pK1 and pK1 can be taken form tables in books for seawater or from a on-line calculator, which I have. A Seawater one is also available but is very complicated for most. I have a calculator programmed for some to these to give CO2 levels in seawater. So, the pH is really a function of CO2, Alk, Salinity, temp, pK1 and pK2. In water, be it FW or salt water, there are 2 pH points , called pK 1 and pK2 and these values are equal to the same pH, i.e pK2 9.1 = pH 9.1. At each one of these two points there is a equal ratio of 1:1 of acid and base, half of one and half of of the other.. If there is a change in salinity or temp these two points change to a different pK1/pH. So, knowing all of this, if your temp drops 1 C we can figure out how much your pH will drop due to the increase in CO2 from that temp drop, which is ~ .01 pH from a temp drop of 1 C. And when the temp rises back up it all shifts again and the pH goes up from the increase in water temp of 1 C and back to or about equal to, your originally pH. Here is an example of the plotted graph. You can see the pK's are at pH ~ 6 and 9. This is for std NSW @ 25 C, 35 ppt. 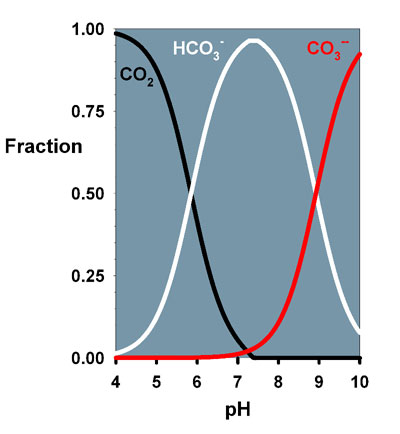 OK, I'll just shut-up 
|
|
#17
|
|||
|
|||
|
Ok that makes much more sense. The only thing I cannot understand is how a temperature drop of 0.4C causes a pH drop of 0.1. You said 1C drop equals approx 0.01 pH drop. so (assuming it is linear) 0.4C should be approx. 0.004pH drop. Why am I seeing such a large drop?
|
|
#18
|
|||
|
|||
|
Oops',
 I made a typo error it is NOT 0.01 pH it is 0.1 pH / 1 C I made a typo error it is NOT 0.01 pH it is 0.1 pH / 1 C  But I did say it here That off-set is enough to drop the pH 0.1 pH units There may be other things to throw it off, such as the meter not really being accurate enough, the temp not being accurate enough, etc..
__________________
If you See Me Running You Better Catch-Up An explosion can be defined as a loud noise, accompanied by the sudden going away of things, from a place where they use to be. Last edited by Boomer; 10/15/2007 at 05:19 PM. |
|
#19
|
|||
|
|||
|
Well the probe has an accuracy of about 0.05 at the most if I have to guess... My Hanna is 0.02 and there is no way the Aquatronica is more accurate. So yes there is a margin of error.
Your "cleared" typo error puts everything in to perspective. Thanks 
|
|
#20
|
|||
|
|||
|
OK

__________________
If you See Me Running You Better Catch-Up An explosion can be defined as a loud noise, accompanied by the sudden going away of things, from a place where they use to be. |
|
#21
|
|||
|
|||
|
Guys, there's no way that a shift in pK1 and pK2 for carbonic acid can explain this.
Yes, pK1 and pK2 both increase with dropping temp, reducing pH, but the effect with 0.5 C of cooling is very small. We also need to consider effects on the pKa of bisulphate, hydrogen fluoride, boric acid, phosphoric acid and silicic acid (though ignoring phosphoric and silicic acid will introduce only a small error in typical surface/reef water) to get a better picture of what happens to pH in sea water. If we consider the solubility of CO2, we should expect slightly more CO2 to go into solution dropping the pH, but again, the difference is very small. If we assume that the pCO2 of the water at equilibrium is 500 uatm (technically we'll use fugacity, which is only a few per mil different) then very slightly more CO2 will dissolve at equilibrium into the cooler water. Assuming S = 35 and an alkalinity of 2300 ueq/kg (which is probably lower than this really is) and the pCO2 is 500 uatm at equilibrum. The calculated pH with these parameters at 27.5 C is: pHnbs = 8.1044 If we maintain the same conditions otherwise but just drop the temperature to 27.0 C and allow the water to come to equilibrium with the atmosphere the calulated pH is: pHnbs = 8.1035 So, a 0.5 C cooling from 27.5 to 27.0 C in sea water, all other things being equal, will cause a 0.0009 reduction in pH. That is a completely immeasurable difference with normal equipment, and I wouldn't put any kind of money on measuring that even with very good equipment. This also doesn't take into account that it does take time for CO2 to dissolve into the water and provide for that drop in pH. If we hold total carbon constant and look at the effect on temperature we see that the pH actually rises: 27.5 C pHnbs = 8.1044 27.0 C pHnbs = 8.1104 So, the immediate effect of cooling (before the water can absorb CO2 and come to equilibrium) is a 0.006 increase in pH, not a reduction. In any event, changes in equilibria of CO2 cannot possibly explain a 0.12 change in pH due to 0.5 C of cooling. That is orders of magnitude bigger than we'd expect. So, the immediate effect of a change in temp near the range of interest is a rise in pH of around 0.01 per 1 C of cooling. The long-term effect is a very, very slight reduction of pH as CO2 is taken up. Chris
__________________
FSM ~ Touched by His noodly appendage ~ Last edited by MCsaxmaster; 10/15/2007 at 09:53 PM. |
|
#22
|
|||
|
|||
|
Is this going to be on the mid-term?
|
|
#23
|
|||
|
|||
|
Yes, and you must calculate everything by hand

__________________
FSM ~ Touched by His noodly appendage ~ |
|
#24
|
|||
|
|||
|
Thanks Chris
 I probably should have not tried to take a short cut based on CO2 and not the actual pH shift from temp  What I did was this pH 8.1, actual carbonate Alk 2.25, pK1 5.966, pK2 9.102 all at 27 C. CO2 = 0.61 mg / l pH 8.0 , actual carbonate Alk 2.25, pK1 5.961, pK2 9.093 all at 28 C. CO2 = 0.78 mg l and then assumed the pH drop would be ~ 0.1 pH for~ 1 C decrease from the 0.17 CO2 increase.
__________________
If you See Me Running You Better Catch-Up An explosion can be defined as a loud noise, accompanied by the sudden going away of things, from a place where they use to be. |
|
#25
|
|||
|
|||
|
Yeah, the whole chemistry can be a bit of a bugger
 Thank goodness for spreadsheets 
__________________
FSM ~ Touched by His noodly appendage ~ |
|
|